Extract Monographs
Rhodiola
Current market production of commercial R. rosea root powder extracts is standardized for the contents of both salidroside and rosavins. But with the limited resources and quantity of output, many Rhodiola P.E. do not come from R. rosea, and are standardized for only salidroside. CPC’s analysis revealed that the quality issue of commercial R. rosea extracts was a great concern because the characteristic bioactive compounds, i.e. rosarin, rosavin and rosin of R. rosea were not detected in 33.3% of the commercial R. rosea extract samples tested. Furthermore, there was also a considerable variation between the actual rosavins content and the amount claimed by suppliers [1].
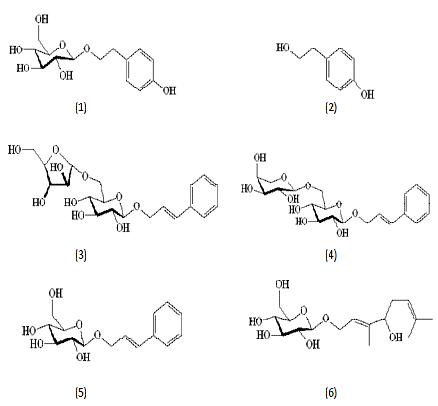
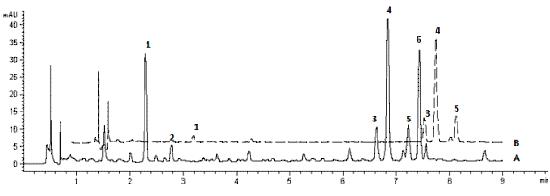
Our Canphy® Rhodiola P.E. is reliable with strict source control and optimal extraction conditions to achieve the highest extraction effectiveness. Meanwhile, our research indicates that the chemical profile of Rhodiola roots does not change during the extraction process. Furthermore, CPC has developed a rapid, sensitive and reliable RRLC method for six biologically active compounds (salidroside, tyrosol, rosarin, rosavin, rosin and rosiridin) in Rhodiola rosea roots and powder extracts conducted within 8 minutes [1]. So, every batch of Canphy® Rhodiola P.E. which is standardized at 3% rosavins is stable and characteristic with the established RRLC profile. In addition, we have developed a RRLC method for polyherbal formulated products with Rhodiola, enabling effective quality control of formulated products containing Rhodiola P.E.[2].
References
[1] Yuan-Chun Ma, et al. Rapid Resolution Liquid Chromatography (RRLC) Analysis for Quality Control of Rhodiola rosea Roots and Commercial Standardized products. Natural Product Communications. 2011, 6(5): 645-650.
[2] Yuan-Chun Ma, et al. Simultaneous quantification of polyherbal formulations containing Rhodiola rosea L. and Eleutherococcus senticosus Maxim. using rapid resolution liquid chromatography (RRLC). Journal of Pharmaceutical and Biomedical Analysis. 2011, 55:908-915.